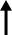|
| A SK Biopharmaceuticals researcher conducts a study for an anti-epileptic drug at the company's research center in Seongnam, Gyeonggi Province, in this file photo. Courtesy of SK Biopharmaceuticals |
By Kim Hyun-bin
SK Biopharmaceuticals has filed a formal application for its share listing on the KOSPI scheduled for June.
The company, a key bio company affiliate of SK Group, submitted a registration statement to the country's financial authorities in the run-up to its initial public offering (IPO), Tuesday.
Shares are expected to start off at an estimated price range between 36,000 won to 49,000 won, which SK Biopharmaceuticals seeks to raise up to 960 billion won ($783 million) by issuing 19.58 million shares to investors.
Local investment agencies are scheduled to meet for two consecutive days starting June 17 to figure out the demand forecast for the soon-to-be-released shares. The following week, subscription process for the shares will be held, which will complete the application procedures to list the company within June.
NH Investment & Securities and Citigroup Global Markets Korea Securities have been selected to oversee the share sale.
SK Holdings fully owns SK Biopharmaceuticals, which was spun off from SK Life Science in 2001.
In November, SK Biopharmaceuticals' anti-epileptic drug XCOPRI won approval from the U.S. Food and Drug Administration (FDA) to treat partial-onset seizures in adults, becoming the first local drug company to receive sales approval from the FDA without exporting its new drug technology.
Industry watchers estimate its corporate value to be at a maximum 5 trillion won.
SK Biopharmaceuticals's IPO is widely projected to be the biggest since the 2017 listing of Netmarble, one of the country's top mobile gaming companies.
The company, established in 1993, has been in charge of research and development of new drugs for 28 years and has been at the forefront in developing medications for diseases involving the central nervous system.
SK Biopharmaceuticals' competitive edge in the global market is its well-established platform to develop new medications as well as its ability to conduct clinical trials and independently commercialize its products in the global market.
The company holds two FDA-approved medications and has been recognized for its technical competitiveness on the international stage.
"The company has developed the country's first epilepsy treatment drug and have independently gained approval by the FDA," an SK Biopharmaceuticals official said. "The new epilepsy drug Cenobamate has been approved by the FDA and has been introduced to the U.S. market this month."
 Subscribe
Subscribe E-Paper
E-Paper