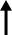|
| President Moon Jae-in, center, poses with Samsung Biologics CEO John Rim, left and Moderna CEO Stephane Bancel during a vaccine partnership event held between Korea and the United States at a hotel in Washington, D.C., May 22. Yonhap |
By Baek Byung-yeul
With Samsung Biologics and U.S. biotechnology firm Moderna agreeing to produce the latter's COVID-19 vaccine at the former's facility in Korea starting in the third quarter of this year, industry officials said Monday the Samsung affiliate was hoping to reach a possible "technology sharing" deal under which it could gain the know-how to independently handle the production of pharmaceutical substances for vaccines.
With President Moon Jae-in's attendance after his summit with Joe Biden in Washington, D.C., Samsung Biologics CEO John Rim inked a "fill and finish" production deal with Moderna, which signifies the filling of vials with drug substances.
Moderna is producing its COVID-19 vaccine using messenger RNA (mRNA) technology. Under the agreement, Samsung and Moderna said "Samsung Biologics will provide large scale, commercial fill-finish manufacturing for mRNA-1273, Moderna's COVID-19 vaccine" at Samsung's production facility in Incheon's Songdo.
Given the country is addressing vaccine shortage issues in line with the government's goal of attaining herd immunity of the population by the end of November this year, the contract would be a plus for Moon and his government. Officials and analysts said because the contract would help expand Samsung's business portfolio, there are chances that Samsung Biologics could be in charge of the entire vaccine production process including producing vaccine substances.
The contract-based vaccine production business consists of drug substance (DS) and drug product (DP) businesses. While Switzerland-based biotechnology firm Lonza is manufacturing drug substances for Moderna's COVID-19 vaccines, SK Securities analyst Lee Dal-mi said Samsung Biologics has "sufficient potential" for producing drug substances as well.
"The company has three manufacturing plants with a fourth under construction. Its total production capacity is 364,000 liters, most of which are for manufacturing antibody drugs. To produce Moderna's mRNA drug substance, Samsung needs to establish an additional facility. Currently, Lonze is producing drug substances for Moderna's vaccine. After constructing additional production facilities, chances are there that Samsung would produce drug substances as well," the analyst said.
An industry official in the local biotechnology industry said striking a manufacturing deal for the Moderna vaccine paves the way for Samsung Biologics' expansion into vaccine production business and the company's business competiveness is expected to be strengthened.
Samsung Biologics started the contract manufacturing organization (CMO) business in 2011 and widened its portfolio into the contract development organization (CDO) business in 2018. CDO is an end-to end biopharmaceutical project running drug development procedures from laboratory research to clinical trials.
"Some say the DP business is a sort of low-hurdle business, but that is not true. It's not a business that anyone can do. As the business deals directly with human life, reliability issues are the most important point in the DP business," the official said on condition of anonymity.
Samsung Biologics is set to begin production of Moderna's COVID-19 vaccine as early as August. Though it usually takes around six months to complete production preparations but the company plans to reduce it to two to three months, according to industry sources.
Regarding the possibility of producing drug substances for Moderna's COVID-19 vaccine, a Samsung Biologics official said, "We cannot confirm the matter as the issue is related to our clients."
 Subscribe
Subscribe E-Paper
E-Paper